The passing of tropical storm, Fay brought some needed precipitation to the region, albeit too much at once in many places. Relatively dry soils (assumes not being over-irrigated) and the long, warm days of summer should allow turfs to dry out reasonably fast. But what about gray leaf spot? Spores of the pathogen are thought […]
Governor Signs Permit Extension Act: NJDEP Water Certifications Extended 1 Year

Governor Murphy signed The Permit Extension Act (A3919/S2346) this week, which extends certain permits during the COVID-19 emergency. Agricultural Certifications issued by the DEP are included in this extension, so any Agricultural Certification which expired on or after March 9, 2020 will be extended.
Any complete new, renewal or modification application already submitted to the DEP will continue to be reviewed and issued for the full five-year term.
Any Certification holder who received the first renewal reminder which contained the pre-filled application form does not need to do anything at this time. The DEP will forward you a new pre-filled application once the extension period is over.
“COVID-19 extension period” means the period beginning March 9, 2020 and continuing for as long as a public health emergency, pursuant to the “Emergency Health Powers Act,” P.L.2005, c.222 (C.26:13-1 et seq.), or a state of emergency, pursuant to P.L.1942, c. 251 (C.App.A.9-33 et seq.), or both, that has been declared by the Governor in response to COVID-19, is in effect.
Please be advised that annual water diversion reports were required to be submitted to the DEP by February 28, 2020 and are not extended due to the passage of this Bill. The DEP is issuing reminders to those who failed to submit this information in accordance with their Certification. Continue to log diversion amounts and submit your reports in accordance with the deadlines in your Certification.
Rutgers Cooperative Extension is working very closely with the DEP’s Bureau of Water Allocation & Well Permitting to resolve any ongoing issues and concerns. The Bureau is aware that many renewal applications are awaiting the Ag Agent review and signature. With the extension act, these renewal applications are being extended as mentioned above.
Some RCE Offices remain closed or with minimal access at this time. Call your local Rutgers Cooperative Extension office for specific information. In addition, during the month of July, staff in the NJ Department of Agriculture and the NJ DEP personnel are furloughed, and response times will be slowed, so please be patient during these challenging times.
Prevailing landscape disease; Entomosporium Leaf Spot on woody ornamentals

Photo: Rich Buckley
It was brought to my attention this morning that many landscapers and plant professionals are concerned by a reddening and spotting discoloration condition on Photinia foliage. Most often this is caused by the fungal pathogen Entomosporium maculatum, a widespread and destructive foliar disease of woody ornamentals in the rose family (including Photinia, Indian Hawthorn, Loquat, flowering and fruiting Pear, Firethorn, Hawthorn, Juneberry, and Quince). This pathogen discolors foliage to an unsightly red, orange, and black spotting that ultimately leads to premature defoliation. The spots will eventually turn necrotic and fungal structures will be visible within those areas (little black dots).

Photo: Rich Buckley
The pathogen can be active from late winter to late fall, favoring conditions that promote humidity and reduced airflow. The pathogen survives on infected leaves (attached or dropped). Typically it takes approximately 2 weeks for symptom development to occur post infection. Therefor the heavy discoloration we are seeing right now is the culmination of multiple infection events, where protectant fungicides were not used or were used correctly.
This means that another round of infections will occur again soon in severely infected locations as the spots will give rise to a new generation of spores.
Disease management (fungicides): Ideally, protectant fungicide applications would have been initiated every 7-14 days post bud break until all leaves had matured. However, in the current situation many people will be playing catch up. Fungicide applications initiated now will not make the symptoms go away or ‘cure’ the plant, but they are required now to protect foliage from the next wave of fungal infections and reduce the overall population. In severely infected plantings, fungicide applications should be continued every 10-14 (depending on fungicide label) days throughout the remainder of the season, especially in high value plantings or in specimen blocks. If winter conditions are unseasonably warm and humid, then applications should continue accordingly. Systemic fungicides should also be considered in addition to protective and cultural control mechanisms in severe cases.
Fungicides:
ROTATE between Fungicide Resistance Action Committee (FRAC codes)
Format: [FRAC code]: Chemical name (Trade names)
- [M05]: Chlorothalonil (Daconil WS)
- [M05 + 1] Chlorothalonil + Thiophanate methyl (Spectro 90WDG)
- [1] Thiophanate methyl (Clearys 3336)
- [11] Azxystrobin (Heritage 50W)
- [3] Myclobutanil (Eagle 40W)
- [3] Propiconazole (Banner Maxx)
- [3] Triadimefon (Bayleton 50, Strike 50WDG)
- [1 + M03] Thiophanate methyl + Mancozeb (Duosan 79W)
- [M03] Mancozeb
- [M01] Copper Oxychloride + Copper Hydroxide (Badge SC, many)
IT IS CRITICAL TO ROTATE BETWEEN FRAC CODES DUE TO THE NUMBER OF POTENTIAL APPLICATIONS
Always refer to the label for allowable host crops, use-restrictions, application rates, reapplication intervals, re-entry intervals (REI), and mix compatibility information.
DISCLAIMER: Production and pesticide information on this site are for commercial operators and landscape professionals only and are NOT for home gardener use. Provided materials represent examples and do not cover all possible control scenarios. Please contact your local agent or chemical sales representative for more information.
Click here for a list of materials available to both homeowners and licensed pesticide applicators
Cultural prevention and mitigation:
- Remove leaf-litter prior to bud break. During the season remove leaf litter only when dry. Infected leaves serve as inoculum for the next season. In severely infected plantings remove and replace mulch as fungal material can be deposited within the litter. Do not use leaf blowers to remove leaf-litter (all that does is spread around the spores).
- Avoid overhead irrigation. Water moving through the canopy represents at least two dangerous plant pathogen situations; increased leaf wetness that lets pathogens infect successfully, and movement of spores (from sporulating leaves and young growth).
- Increase airflow. Like all plant diseases, any situation that increases airflow, will decrease leaf wetness. This could include pruning, decreasing the density of plants, greater spacing, and planting/rearing site selection.
- Pruning in heavily infected plants. In addition to promoting increased air flow, heavy pruning may be required to remove overwintering inoculum from the plants. This should only be done during dry conditions, preferably during plant dormancy periods.
For sample identification please contact the Plant Diagnostic Lab here at Rutgers.

Photo: Rich Buckley
For more information and tips please visit:
Controlling Entomosporium Leaf Spot on Woody Ornamentals
Photinia Leaf Spot – Prevention And Treatment Of Common Photinia Bush Diseases
Entomosporium Leaf spot of Photinia and Indian Hawthorn
Entomosporium Leaf Spot Diagnostic Guide
Timothy J. Waller, Ph.D. (twaller@njaes.rutgers.edu)
Boxwood blight update for 6/22-6/26/20
There are consistently high infection potentials this week for boxwood blight.
Protective fungicide applications should remain in effect or be initiated immediately.
| Boxwood Blight Risk Model | ||||
| Date | Upper Deerfield (NJ50) | Howell (NJ10) | Pequest (NJ58) | |
| 6/20/20 | S | Low risk | Very low risk | Infection potential |
| 6/21/20 | Sun | Low risk | Infection potential | Infection potential |
| 6/22/20 | M | Infection potential | Infection potential | Infection potential |
| 6/23/20 | T | Infection potential | Infection potential | High Inf. potential |
| 6/24/20 | W | High Inf. potential | High Inf. potential | High Inf. potential |
| 6/25/20 | Th | Infection potential | Infection potential | Low risk |
| 6/26/20 | F | Very low risk | Very low risk | Low risk |
| — | — | — | — | — |
| ^Risk of infection^ | ||||
| adapted from: uspest.org/risk/boxwood_app | ||||
The weather: Don’t let these beautiful early summer days fool you, we all know that violent thunderstorms can appear out of nowhere, bringing with them high winds and rain (wind driven rain) causing high persistent humidity. I.e. the perfect storm for fungal pathogens to spread and successfully infect. Accordingly, many other fungal pathogens are equally active under these conditions and should be managed accordingly. Prevention is always easier than a cure.
These advisories are general in nature so someone from your business should be using this risk model if boxwood is important to your financial stability. In 30 seconds you can have a better idea of boxwood blight (and other pathogen) activity in your immediate area!
Boxwood blight information:
The disease may be sporulating and spreading amongst plantings if present (or may come into your nursery or landscapes via shipments) within the next few days. The pathogen, Calonectria pseudonaviculata, (aka Cylindrocladium buxicola) can also infect other hosts within the boxwood family (Buxaceae). These include common landscape plants such as Pachysandra and Sweetbox, so treatments should be directed towards these hosts as well. Leaf-litter suspected of boxwood blight should only be removed carefully under dry conditions (i.e. do not use a leaf blower to remove leaf-litter in Pachysandra or Boxwood plantings this week!). Additionally, please refrain from touching the plants at all during wet conditions as boxwood blight spores are sticky and can be transferred from plant-to-plant or site-to-site. Please follow proper sanitization and sterilization practices in addition to a preventative spray program. Tools, clothing, and transportation equipment should be routinely cleaned with alcohol or bleach to prevent the spread of this disease. Additionally, this pathogen forms small (yet bomb-proof) survival structures (Microsclerotia) that can remain as a source of infections for 7-10 years within the soil and leaf-litter (part of why you do not want to use a leaf blower to clean out leaf-litter!).
See the following for what to do in multiple boxwood blight scenarios.
Boxwood blight key symptoms:
- Straw-yellow to bronzing leaves that have a characteristic halo of lighter brown and yellow around the infection point.
- LEAVES FALL OFF, TYPICALLY STARTING AT THE BOTTOM. These leaves then act as a source for new infections.
- Twigs will have long brown lesions
Look-a-likes: NOT ALL FOLIAR DISEASES are Boxwood blight:
- Volutella Blight (straw-yellow or golden leaves with pink spores, branches have loose bark).
- Macrophoma leaf spot (straw-yellow leaves with black fungal structures (black spots))
Fungicides;
ROTATE between Fungicide Resistance Action Committee (FRAC codes)
Format: [FRAC code]: Chemical name (Trade names)
- [M05]: Chlorothalonil (Daconil WS)
- [M05 + 1] Chlorothalonil + Thiophanate methyl (Spectro 90WDG)
- [11] Trifloxystrobin + [7] Fluopyram (Broadform)
- [11] Trifloxystrobin + [3] Triadimefon (Armada 50WDG)
- [M03] Mancozeb
- [12] Fludioxonil (Medallion WDG)
- [3] Tebuconazole (Torque)
Always refer to the label for allowable host crops, use-restrictions, application rates, reapplication intervals, re-entry intervals (REI), and mix compatibility information.
DISCLAIMER: Production and pesticide information on this site are for commercial operators only and are NOT for home gardener use. Provided materials represent examples and do not cover all possible control scenarios. Please contact your local agent or chemical sales representative for more information.
Previous Boxwood Blight Plant and Pest Advisory Posts;
- Immediate potential for boxwood blight activity (by Tim Waller)
- Boxwood blight revisited (by Rich Buckley)
- Boxwood blight confirmed in New Jersey (by Rich Buckley)
Additional resources on boxwood blight;
- NC State: Effective materials for boxwood blight
- Virginia Tech extension: Cultural practices and resistant varieties
- Perdue Extension boxwood blight
- University of Kentucky Extension boxwood blight
- Nursery Magazine “Reclaiming boxwood from boxwood blight”
- NJ Department of Agriculture boxwood blight guidelines
Contact: Timothy J. Waller, Ph.D. (twaller@njaes.rutgers.edu)
Nursery update; focus on warm season spider mites
This advisory provides; (1) warm season spider mite biology and control options, (2) current growing degree-day information, and (3) a boxwood blight risk update.
Note: Previous spider mite advisories
Steve Rettke, Rutgers IPM, wrote two very good advisory posts pertaining to the different seasonal mites, in addition to symptomology and images of the different mites. He is a wealth of information and has contributed photos and will be assisting in future fact-sheets pertaining to common pests and their control. In light of these advisory posts (below) this current advisory will only cover the basics as it applies to pest control. Contact: rettke@njaes.rutgers.edu
Please review his very informative advisories:
Cool season mites: https://plant-pest-advisory.rutgers.edu/cool-season-mites-wax-as-the-warm-season-mites-wane/
Warm season mites: https://plant-pest-advisory.rutgers.edu/cool-season-mites-have-faded-but-warm-season-mites-now-surge/

Cool season: Spruce spider mite damage on an Aberta Dwarf Spruce. Photo: Steven Rettke, RCE.

Warm season: Two spotted spider mite webbing and damage. Photo: Steven Rettke, RCE.
(2) Basic spider mite biology and control options
Cool season mites: The mites affecting horticulture crop production can be broken into two functional groups; cool season and warm season. Cool season mites feed and lay eggs, as the name suggests, in cooler times of the year (late winter-early spring and then again fall-early winter depending on the year). The two most common cool season mites are the Spruce and Southern red spider mites that feed on conifers and broadleaf evergreens, respectively. In many cases the damages caused by these mites are not noticed until well into the growing season. Meaning that if damages are observed mid-season you would be better off waiting until the fall to control cool season mites, i.e. targeting late season egg hatches and developing mites.
Warm season mites: As the season progresses towards summer, the warm season mites including Two-spotted, Maple, Oak, Honey locust, European, and other host specific spider mites become active (typically June – September). The Two-spotted spider mite is known as a “cosmopolitan” pest because of its ability to colonize, and damage, a wide variety of horticulture crops (but are rarely observed on conifers). Sentinel plants would include rose, euonymus, and forsythia, as they are favorites of these mites. In contrast to the cool season mites, warm season mite damages are often visible while the pest is actively feeding and multiplying, as up to 10-15 generations per growing season can impact your crop. Warm season mites evade suffocation by dormant oil sprays and other miticide applications by leaving their feeding locations on plants once temperatures begin to drop, and overwinter in the soil horizon or leaf litter under the host plant. As warm season mites are preparing to go into dormancy they begin to change their color to an orange hue, meaning they are not feeding as readily and will not be as affected by miticides because of this.

Multiple life stages of Two spotted spider mites. Photo: Steven Rettke, RCE.

Two spotted spider mite females going into dormancy (orange coloration). Photo: Steven Rettke, RCE.
Conditions favoring spider mites: Full sun and dry, dusty conditions are often associated with increasing mite populations as low relative humidity often stimulates mite reproduction. Additionally, during periods of rain (and resulting leaf wetness-periods), certain fungi are able to attack and reduce mite populations (called entomopathogenic fungi). Meaning in frequently wet seasons mite numbers, damage, and miticide applications should be lowered due to increased fungal activity. However, this situation can certainly lead to increased plant pathogenic fungi activity as well, therefor a balance between the two is most desirable. Excessive nitrogen fertilizer has also been linked to explosive mite populations; therefor a well-planned nutritional regiment should be strictly followed.
IPM is important when suppressing spider mite populations!
Beneficials: In addition to entomopathogenic fungi (like Beauveria bassiana), numerous beneficial organisms can also manage mite populations including predacious mites (Phytoseiidae family), spider mite destroyer (Stethorus punctillim), ladybug larvae, midge larvae (Feltiella acarisuga), and minute pirate bugs. Given this complex, and many times free, natural defense system, applications of certain insecticides can actually cause mite populations to resurge by killing the mite-feeding beneficials (see note below). Therefor, all applications should be thoughtfully considered with economic (or cosmetic) thresholds in mind.
Compounds that can cause spider mite resurgence:
- Carbaryl (Sevin)
- Cyfluthrin containing compounds (Tempo, Decathalon, Scimitar)
- Permethrin (Astro)
- Imdacloprid (Merit, Marathon)
Warm season spider mite treatment options:
ROTATE between Insecticide Resistance Action Committee (IRAC #)
Format: [IRAC Group #]: Chemical name (Trade names)
- [3A]: Pyrethrins (Tersus)
- [5]: Spinosad (Conserve, Entrust)
- [6]: Abamectin (Avid, Lucid, Aracinate TM)
- [20D]: Bifenazate (Floramite SC)
- [10A]: Hexythiazox (Hexygon)
- [21A]: Pyridaben (Sanmite, Sanmite SC)
- [21A]: Fenazaquin (Magus)
- [23]: Spiromesifen (Forbid 4F, Judo)
“Softer” spider mite materials
- Agitator (forces mites out of hiding): Captiva
- Azadirachtin from the Neem plant: (Azatrol EC)
- Beauveria bassiana: (Naturalis-L)
- Horticulture oil: (@ 1-2% vol:vol summer applications)
- Insecticidal soaps: (M-Pede)
Always refer to the label for allowable host crops, use-restrictions, application rates, reapplication intervals, REI times, and mix compatibility information.
NOTE: Production and pesticide information on this site are for commercial operators only and are NOT for home gardener use. Provided materials represent examples and do not cover all possible control scenarios. Please contact your local agent or chemical sales representative for more information.
(2) Growing Degree-days as of 6/16/2020 (if you need help please contact me at twaller@njaes.rutgers.edu)
Upper Deerfield (NJ50)= 809 GDD50 Howell/Freehold (NJ10) = 628 GDD50 Pequest (NJ58) = 525 GDD50
Growing degree day map for our area: (courtesy of Cornell University; Fore Cast)
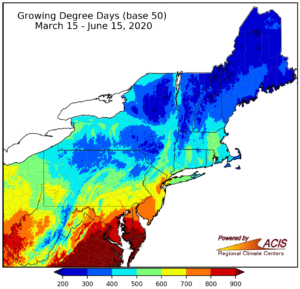
Growing degree-day map for our area. Cornell, Fore Cast.
(3) Boxwood blight risk update
| Boxwood Blight Risk Model | ||||
| Date | Upper Deerfield (NJ50) | Howell (NJ10) | Pequest (NJ58) | |
| 6/14/20 | Sun | Very low risk | Very low risk | Very low risk |
| 6/15/20 | M | Very low risk | Very low risk | Very low risk |
| 6/16/20 | T | Very low risk | Very low risk | Very low risk |
| 6/17/20 | W | Very low risk | Very low risk | Very low risk |
| 6/18/20 | Th | Low risk | Low risk | Low risk |
| 6/19/20 | F | Infection potential | Infection potential | Low risk |
| 6/20/20 | S | Low risk | Infection potential | Low risk |
| 6/21/20 | Sun | Very low risk | Low risk | Very low risk |
| Risk of infection | ||||
| adapted from: https://uspest.org/risk/boxwood_app | ||||
Please visit this website (there is a mobile app as well): https://uspest.org/risk/boxwood_app
Protective fungicide applications should remain in effect or be initiated immediately for Boxwood Blight. The disease may be sporulating and spreading amongst plantings if present (or may come into your nursery via shipments) within the next few days. Please continue preventative/protectant fungicide applications utilizing; chlorothalonil (FRAC Group M05), mancozeb (FRAC Group M03), and QoI-fungicides (FRAC Group 11) in a rotation (other materials available as well).
NJAES; Plant Diagnostic Lab https://njaes.rutgers.edu/plant-diagnostic-lab/
*Additional links and resources*
Please take the 2020 Nursery Industry Survey: https://forms.gle/dUjLxaiu6qDQYYsRA
Ask the Agent (every Wednesday @ 7:00pm): : https://go.rutgers.edu/t7wjkit1
NJNLA- Wine Down Wednesdays (every first and third Wednesday @ 7:00pm-8:00pm).
Email: Lori Jenssen @ njnla.director@gmail.com
Subscribe to the NJAES YouTube webpage: https://www.youtube.com/user/RutgersNJAES/
RU Sustainable Nursery Production website: https://njaes.rutgers.edu/nursery/
March meeting PDF: https://njaes.rutgers.edu/nursery/documents/2020%20SJ%20Nursery%20Meeting%20Proceedings.pdf
How to use growing degree-day resources: https://plant-pest-advisory.rutgers.edu/using-growing-degree-day-calculators-nursery-pest-and-pathogen-updates-important-resource-links/
2017 Southeastern U.S. Pest Control Guide for Nursery Crops and Landscape Plantings: https://content.ces.ncsu.edu/southeastern-us-pest-control-guide-for-nursery-crops-and-landscape-plantings
Rutgers Insect Control Recs. for Shade Trees and Commercial Nursery Crops: https://njaes.rutgers.edu/pubs/publication.php?pid=E334
NJAES; Soil Testing Lab: https://njaes.rutgers.edu/soil-testing-lab/
Thank you,
Cumberland County Extension: Timothy J. Waller, Ph.D. @ twaller@njaes.rutgers.edu
Scale insect crawler activity and summer treatment options
Basic Scale Biology
There are two types of scale insects; armored and soft scales. Scale insects hatch from eggs and typically develop through 2 nymphal instar growth stages known as “crawlers”. During this “crawler stage” (many cases mid-May through July) the insects are vulnerable to insecticides and other biological enemies. Once the “crawlers” settle on a feeding location and begin to develop waxy or hard outer coverings, the insects are no longer susceptible to most insecticides.
Targeting the “crawler” stage is imperative to successful pest management.
One slight difference between armored and soft scales is how many generations they can have per year; armored (several), soft (typically only 1). Also, once settled armored scales are stationary whereas soft scales can move (albeit slowly). Many scale insect “crawlers” head towards growing vegetative tips, making this location suitable for scouting. Various sticky traps, including double sided tape wrapped around branches are utilized to determine the presence of scale “crawlers”. As noted prior, the presence of “crawlers” should be first visually confirmed if at all possible as pesticide applications made too early are not effective. Programs that utilized cool season dormant/horticultural oils, proper pruning, and the use of summertime contact or systemic insecticides are most successful.(A detailed scale insect fact-sheet is forthcoming!)
Armored and soft scales can be controlled using the same chemicals if targeting “crawlers”.
Growing Degree-days as of 6/10/2020 (Weather station selected for each location. Ex: NJ50 = Upper Deerfield)
Upper Deerfield (NJ50)= 686 GDD50 Howell/Freehold (NJ10) = 536 GDD50 Pequest (NJ58) = 441 GDD50
Scale insects target growing degree-days
(Egg hatch + crawler emergence = scout and treat as needed)
Pine needle scale – (448)
Maskell scale – (470)
Oystershell scale – (486)
Cottony camellia/taxus scale – (520)
Euonymus scale – (522)
European elm scale – (542)
White prunicola (Peach) scale – (594)
Cryptomeria scale – (600-800)
Golden oak scale (680)
Juniper scale – (694)
Hemlock ‘fioriniae’ scale – (300-799)
Spruce bud scale – (700)
Fletcher Scale – (730)
Azalea bark scale – (737)
Oak lecanium scale – (789)
Beech scale – (800)
Japanese maple scale – (829)
Calico scale – (834)
Cottony maple scale – (850)
European fruit lecanium scale – (940 DD)
Summertime scale treatment options
ROTATE between Insecticide Resistance Action Committee (IRAC #)
Format: [IRAC Group #]: Chemical name (Trade names)
- [1A]: Carbaryl (Sevin SL)
- [1B]: Acephate (Orthene, Leptitect, Precise GN)
- [1B]: Chlorpyrifos (Dursban 50W, DuraGuard ME)
- [1B]: Malathion (Malathion 5 EC)
- [3A]: Cyfluthrin (Decathlon)
- [3A]: Bifenthrin (Talstar S Select, Onyx)
- [3A]: lambda-cyhalothrin (Scimitar GC)
- [3A + 4A] Imidacloprid + Bifenthrin (Allectus SC)
- [3A + 4A] Imidaclopird + cyfluthrin (Discuss NG)
- [4A]: Acetamiprid (TriStar 8.5 SL)
- [16]: Buprofezin (Talus 70DF)
- [7C]: Pyriproxifen (Distance IGR)
- Insecticidal soaps (M-Pede)
- Azadirachtin from the Neem plant: (Aza-Direct, Azatin O or XL, Azatrol EC)
Please read the label. The label will provide allowable plant species, use rates, restrictions (phytotoxicity or pollinator concerns), and re-application intervals.
Some useful scale resources: (bare in mind there is variability amongst models and GDD depending on the source, but generally the information will help you make better pest management choices)
- https://www.canr.msu.edu/uploads/files/e2812.pdf
- https://plant-pest-advisory.rutgers.edu/beware-of-the-cryptomeria-scale-hiding-beneath-a-transparent-cover/
- https://www.canr.msu.edu/news/managing_soft_scales_in_conifers#:~:text=Spruce%20bud%20scale%20is%20typically,more%20often%20than%20higher%20branches.&text=Usually%2C%20eggs%20hatch%20and%20tiny,or%20GDD%2C%20base%205).
- http://cues.cfans.umn.edu/old/Web/102CottonyMapleScale.pdf
- https://www.canr.msu.edu/ipm/agriculture/christmas_trees/gdd_of_landscape_insects
- https://plant-pest-advisory.rutgers.edu/beware-of-the-cryptomeria-scale-hiding-beneath-a-transparent-cover/
Southeastern U.S. Pest Control Guide for Nursery Crops and Landscape Plantings: https://content.ces.ncsu.edu/southeastern-us-pest-control-guide-for-nursery-crops-and-landscape-plantings
Rutgers Insect Control Recommendations for Shade Trees and Commercial Nursery Crops: https://njaes.rutgers.edu/pubs/publication.php?pid=E334
Online events and Rutgers Resources:
Ask the Agent (every Wednesday @ 7:00pm): Join: https://go.rutgers.edu/t7wjkit1
NJNLA- Wine Down Wednesdays (every first and third Wednesday @ 7:00pm-8:00pm). Email: Lori Jenssen @ njnla.director@gmail.com
Please take the 2020 Nursery Industry Survey: https://forms.gle/dUjLxaiu6qDQYYsRA
Subscribe to the NJAES YouTube webpage: https://www.youtube.com/user/RutgersNJAES/
RU Sustainable Nursery Production website: https://njaes.rutgers.edu/nursery/
How to use growing degree-day resources: https://plant-pest-advisory.rutgers.edu/using-growing-degree-day-calculators-nursery-pest-and-pathogen-updates-important-resource-links/
NJAES; Plant Diagnostic Lab: https://njaes.rutgers.edu/plant-diagnostic-lab/
NJAES; Soil Testing Lab: https://njaes.rutgers.edu/soil-testing-lab/
Contact: Timothy Waller, Ph.D. at: twaller@njaes.rutgers.edu
